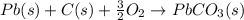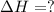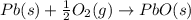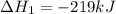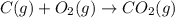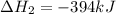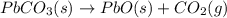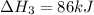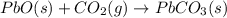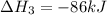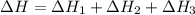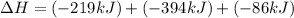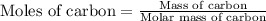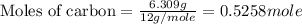Chemistry, 23.10.2019 19:30 desiiraee6265
Use the following information to calculate the amount of heat involved in the complete reaction of 6.309 of carbon to from pbco3 (s) in reaction 4. be sure to give the proper sign (positive or negative) with your answer. (1) pb(s)+1/2 o2 right arrow pbo(s) (2) c(g)+o2(g) right arrow co2(g) (3) pbco3(s)right arrow pbo(s)+co2(g) (4) pb(s)+c(s)+3/2 o2(g) right arrow pbo3(s) delta h degree rsn= -219 kj delta h degree rsn= -394 kj delta h degree rsn= 86 kj

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:30
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1

Chemistry, 22.06.2019 12:30
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1


You know the right answer?
Use the following information to calculate the amount of heat involved in the complete reaction of 6...
Questions




Computers and Technology, 19.09.2019 18:20



Computers and Technology, 19.09.2019 18:20










English, 19.09.2019 18:20

Physics, 19.09.2019 18:20


Mathematics, 19.09.2019 18:20