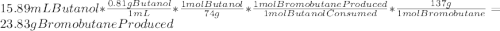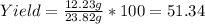1-butanol yields 1-bromobutane in the presence of concentrated sulfuric acid and an excess of sodium bromide. ch3ch2ch2ch2oh (l) → ch3 ch2ch2ch2br (l) if 15.89 ml of 1-butanol produced 12.23 g of 1-bromobutane, the percentage yield of the product equals: (assume the density of 1-butanol is 0.81 g/ml, the molar mass of 1-butanol is 74 g/mol, and the molar mass of 1-bromobutane is 137 g/mol.)

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 17:30
What is the formula for the molecular compound nitrogen monoxide
Answers: 1

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 10:30
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2

Chemistry, 22.06.2019 16:00
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1
You know the right answer?
1-butanol yields 1-bromobutane in the presence of concentrated sulfuric acid and an excess of sodium...
Questions




Biology, 16.05.2021 06:30

Social Studies, 16.05.2021 06:30


Biology, 16.05.2021 06:30

Chemistry, 16.05.2021 06:30



Mathematics, 16.05.2021 06:30



Health, 16.05.2021 06:30

History, 16.05.2021 06:30



Mathematics, 16.05.2021 06:40


Mathematics, 16.05.2021 06:40