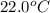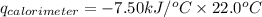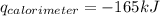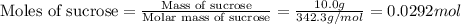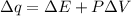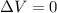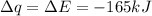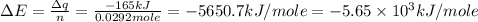Consider the reaction c12h22o11(s)+12o2(g)→12co2(g)+11h2o (l) in which 10.0 g of sucrose, c12h22o11, was burned in a bomb calorimeter with a heat capacity of 7.50 kj/∘c. the temperature increase inside the calorimeter was found to be 22.0 ∘c. calculate the change in internal energy, δe, for this reaction per mole of sucrose. express the change in internal energy in kilojoules per mole to three significant figures.

Answers: 2
Another question on Chemistry

Chemistry, 23.06.2019 06:30
Acertain atom has 22 protons and 19 electrons. this atom loses an electron. the net charge on the atom is now 4+1+01-4-. if this same atom with 22 protons and 19 electrons were to gain 3 electrons, the net charge on the atom would be 3+2+02-3-.
Answers: 1

Chemistry, 23.06.2019 07:00
Choose the correct statement about licensed veterinarians in the united states. a. they must be certified by the avma. b. they can treat all nonhuman animals. c. they can can treat only animals specified on the license. d. they must choose a specialty.
Answers: 2

Chemistry, 23.06.2019 10:00
What is the density, d, of a substance with a volume of v = 12.5 cm3 and a mass of m = 74.4 g ?
Answers: 1

Chemistry, 23.06.2019 10:00
How to draw a diagram to represent a calcium metal lattice?
Answers: 3
You know the right answer?
Consider the reaction c12h22o11(s)+12o2(g)→12co2(g)+11h2o (l) in which 10.0 g of sucrose, c12h22o11,...
Questions


Health, 10.10.2019 10:00

History, 10.10.2019 10:00

English, 10.10.2019 10:00




Social Studies, 10.10.2019 10:00


Chemistry, 10.10.2019 10:00



English, 10.10.2019 10:00

Health, 10.10.2019 10:00

Chemistry, 10.10.2019 10:00




Physics, 10.10.2019 10:00

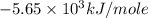
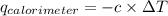
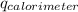 = heat released by calorimeter = ?
= heat released by calorimeter = ?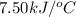
 = change in temperature of calorimeter =
= change in temperature of calorimeter =