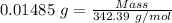Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 20:20
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1

Chemistry, 22.06.2019 23:00
If two identical atoms are bonded,what kind of molecule is formed
Answers: 1
You know the right answer?
Atetraphenyl phosphonium chloride (tppcl) powder (fw=342.39) is 94.0 percent pure. how many grams ar...
Questions


Mathematics, 26.01.2021 03:20

Mathematics, 26.01.2021 03:20



History, 26.01.2021 03:20



Mathematics, 26.01.2021 03:20

Chemistry, 26.01.2021 03:20

Geography, 26.01.2021 03:20


Mathematics, 26.01.2021 03:20




Computers and Technology, 26.01.2021 03:20

Health, 26.01.2021 03:20

Mathematics, 26.01.2021 03:20

History, 26.01.2021 03:20