
Chemistry, 19.10.2019 04:20 viktoria1198zz
If the percent yield for the following reaction is 80.37%, and 63.21 g of no2 are used in the reaction mixture, how many grams of nitric acid, hno3(aq), are produced in the experiment? 3 no2(g) + h2o(l) → 2 hno3(aq) + no(g) molar mass no2 = 46.01 g/mol and hno3 = 63.01 g/mol.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 13:30
Which of the following natural processes is most likely to support the formation of an underwater sinkhole? a pollution buildup from deposited minerals b limestone cave collapsing due to changes in sea level c erosion of large amounts of sand moved by ocean waves d oxidation of rock formed by chemical weathering
Answers: 1

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 15:30
How does a large body of water, such as the ocean, influence climate?
Answers: 1
You know the right answer?
If the percent yield for the following reaction is 80.37%, and 63.21 g of no2 are used in the reacti...
Questions

History, 18.03.2021 03:00



Mathematics, 18.03.2021 03:00

Mathematics, 18.03.2021 03:00


Mathematics, 18.03.2021 03:00

Physics, 18.03.2021 03:00


Mathematics, 18.03.2021 03:00




Mathematics, 18.03.2021 03:00


Mathematics, 18.03.2021 03:00

Advanced Placement (AP), 18.03.2021 03:00

Mathematics, 18.03.2021 03:00


Mathematics, 18.03.2021 03:00

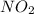 is 46.01 g/mol and mass of
is 46.01 g/mol and mass of 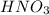 is 63.01 g/mol.
is 63.01 g/mol.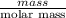
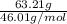
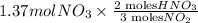
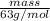
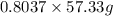
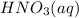 , are produced in the experiment.
, are produced in the experiment.

