
This question has multiple parts.
a. in a coffee-cup calorimeter, 1.81 g nh4no3 was mixed with 85.00 g water at an initial temperature 25.00oc. after dissolution of the salt, the final temperature of the calorimeter contents was 23.34oc.
assuming the solution has a heat capacity of 4.18 j/g∙oc, and assuming no heat loss to the calorimeter, calculate the enthalpy of solution (∆hsoln) for the dissolution of nh4no3 in units of kj/mol.
∆ = kj/mol
b. in a coffee-cup calorimeter, 1.81 g nh4no3 was mixed with 85.00 g water at an initial temperature 25.00oc. after dissolution of the salt, the final temperature of the calorimeter contents was 23.34oc.
if the enthalpy of hydration for nh4no3 is -630. kj/mol, calculate the lattice energy of nh4no3.
lattice energy = kj/mol

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
If a bottle of olive oil contains 1.4 kg of olive oil, what is the volume, in milliliters ( ml ), of the olive oil?
Answers: 1

Chemistry, 22.06.2019 07:30
Compare and contrast the bohr model and the electron cloud models of the atom.
Answers: 1

Chemistry, 22.06.2019 10:00
Diffraction is when light is bent around obstructions. which of the these observation about clouds would indicate diffraction? a) after rain storms, you can sometimes see rainbows. b) clouds are white or gray and cannot be seen through. c) on a cloudy day, the temperature tends to be cooler than a sunny day. d) the edges of dark clouds appear lighter. this
Answers: 3

Chemistry, 22.06.2019 14:00
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2
You know the right answer?
This question has multiple parts.
a. in a coffee-cup calorimeter, 1.81 g nh4no3 was mixed with...
a. in a coffee-cup calorimeter, 1.81 g nh4no3 was mixed with...
Questions

Mathematics, 09.07.2019 18:00

History, 09.07.2019 18:00


History, 09.07.2019 18:00

Mathematics, 09.07.2019 18:00

History, 09.07.2019 18:00

History, 09.07.2019 18:00

Mathematics, 09.07.2019 18:00


Mathematics, 09.07.2019 18:00

Mathematics, 09.07.2019 18:00

Mathematics, 09.07.2019 18:00

History, 09.07.2019 18:00

History, 09.07.2019 18:00

Chemistry, 09.07.2019 18:00

Mathematics, 09.07.2019 18:00

Mathematics, 09.07.2019 18:00


Mathematics, 09.07.2019 18:00

Health, 09.07.2019 18:00

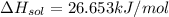

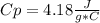
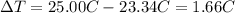
![m=m_{water} + m_{salt]=1.81g +85g= 86.81g](/tpl/images/0333/2995/cfc86.png)
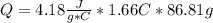
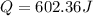
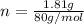
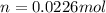
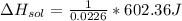
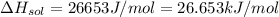
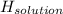 = 26.20 kJ/mol
= 26.20 kJ/mol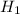 ) = - 656.20 kJ/mol
) = - 656.20 kJ/mol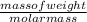
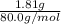

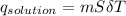
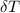 = 25.00°C - 23.34°C
= 25.00°C - 23.34°C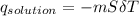 (since heat is lost by the water to the compound)
(since heat is lost by the water to the compound)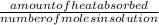
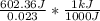
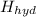 + Δ
+ Δ

