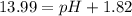Chemistry, 18.10.2019 01:30 DragonWarrior203
Determine the ph of the following base solutions. (assume that all solutions are at 25°c and the ion-product constant of water, kw, is 1.01 ✕ 10−14.) (a) 1.39 ✕ 10−2 m naoh webassign will check your answer for the correct number of significant figures. (b) 0.0051 m al(oh)3 webassign will check your answer for the correct number of significant figures.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:10
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2



Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1
You know the right answer?
Determine the ph of the following base solutions. (assume that all solutions are at 25°c and the ion...
Questions



Mathematics, 28.01.2020 08:31


English, 28.01.2020 08:31


Mathematics, 28.01.2020 08:31

Mathematics, 28.01.2020 08:31

Chemistry, 28.01.2020 08:31





Mathematics, 28.01.2020 08:31



History, 28.01.2020 08:31

Spanish, 28.01.2020 08:31

Mathematics, 28.01.2020 08:31

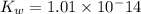
![K_w=[H^+][OH^-]](/tpl/images/0330/0905/bc68a.png)
![1.01\times 10^-{14}=[H^+][OH^-]](/tpl/images/0330/0905/ef4b3.png)
![-\log[1.01\times 10^-{14}]=(-\log [H^+])+(-\log [OH^-])](/tpl/images/0330/0905/fdeff.png)
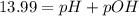
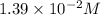 of NaOH.
of NaOH.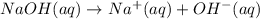
![[OH^-]=1\times [NaOH]=1\times 1.39\times 10^{-2} M=1.39\times 10^{-2} M](/tpl/images/0330/0905/52ef4.png)
![pOH=-\log[1.39\times 10^{-2} M]=1.86](/tpl/images/0330/0905/dac37.png)
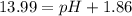
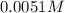 of NaOH.
of NaOH.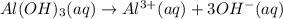
![[OH^-]=3\times [Al(OH)_3]=3\times 0.0051 M=0.0153 M](/tpl/images/0330/0905/db0a1.png)
![pOH=-\log[0.0153 M]=1.82](/tpl/images/0330/0905/47516.png)