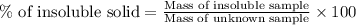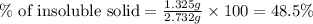Chemistry, 18.10.2019 01:00 sierravick123owr441
What is the percentage of insoluble sample in the unknown if the original sample had a mass of 2.732 g and that of the insoluble solid was determined to be 1.325 g? don't include the % sign in your answer.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 13:50
Aluminum–lithium (al-li) alloys have been developed by the aircraft industry to reduce the weight and improve the performance of its aircraft. a commercial aircraft skin material having a density of 2.47 g/cm3 is desired. compute the concentration of li (in wt%) that is required.
Answers: 3

Chemistry, 22.06.2019 13:00
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1


Chemistry, 22.06.2019 18:00
Many pharmaceutical drugs are organic compounds that were originally synthesized in the laboratory. which two scientific disciplines are bridged by pharmaceutical drugs? chemistry and forensics chemistry and medicine biology and forensics biology and criminology
Answers: 2
You know the right answer?
What is the percentage of insoluble sample in the unknown if the original sample had a mass of 2.732...
Questions

Health, 02.03.2021 04:50

Mathematics, 02.03.2021 05:00


Biology, 02.03.2021 05:00

English, 02.03.2021 05:00

English, 02.03.2021 05:00

Mathematics, 02.03.2021 05:00


Mathematics, 02.03.2021 05:00

Mathematics, 02.03.2021 05:00

English, 02.03.2021 05:00


Mathematics, 02.03.2021 05:00


Mathematics, 02.03.2021 05:00

Mathematics, 02.03.2021 05:00


Mathematics, 02.03.2021 05:00

English, 02.03.2021 05:00