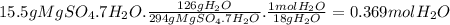Chemistry, 16.10.2019 01:30 Serenitybella
Bath salts such as epsom contain hydrate salts such as mgso 4 ·7h 2 o (magnesium sulfate heptahydrate). when heated, water is released as vapor and anhydrous mgso 4 remains. determine how many moles of vapor are released when 15.5 g of mgso 4 ·7h 2 o are heated. hint: include h 2 o when calculating the molar mass.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:40
Determine the energy released per kilogram of fuel used. given mev per reaction, calculate energy in joules per kilogram of reactants. consider 1 mole of tritium plus 1 mole of deuterium to be a mole of “reactions” (total molar mass = 5 grams).
Answers: 1

Chemistry, 22.06.2019 09:00
Identify the electromagnets with poles that are reversed from the electromagnet shown above
Answers: 3

Chemistry, 22.06.2019 14:30
Which of the following describes a situation where competition between producers exists
Answers: 1

You know the right answer?
Bath salts such as epsom contain hydrate salts such as mgso 4 ·7h 2 o (magnesium sulfate heptahydrat...
Questions

SAT, 21.01.2021 17:20




History, 21.01.2021 17:20




Chemistry, 21.01.2021 17:20




English, 21.01.2021 17:20





History, 21.01.2021 17:20


Chemistry, 21.01.2021 17:20