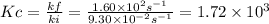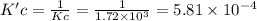At a given temperature, the elementary reaction a > b in the forward direction is first order in a with a rate constant of 1.60*10^2 s^-1. the reverse reaction is first order in b and the rate constant is 9.30*10^-2 s^-1what is the value of the equilibrium constant for the reaction a > b at this temperature? what is the value of equilibrium constant for the reaction b--> a at this temperature?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Asap! how do you lengthen a pattern piece? (family and consumer science, sewing)
Answers: 1

Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1


Chemistry, 22.06.2019 16:00
Click the button that shows the correct relationship of the electron affinities of the elements sodium and phosphorus. sodium’s electron affinity value is more negative than the electron affinity value of phosphorus. phosphorus’ electron affinity value is more negative than the electron affinity value of sodium. this information cannot be determined using the periodic table. answer is b on e2020.
Answers: 3
You know the right answer?
At a given temperature, the elementary reaction a > b in the forward direction is first order in...
Questions

Biology, 05.10.2020 23:01


Chemistry, 05.10.2020 23:01


Social Studies, 05.10.2020 23:01

History, 05.10.2020 23:01

Spanish, 05.10.2020 23:01

Health, 05.10.2020 23:01

English, 05.10.2020 23:01

History, 05.10.2020 23:01

Chemistry, 05.10.2020 23:01



English, 05.10.2020 23:01





Geography, 05.10.2020 23:01

Mathematics, 05.10.2020 23:01