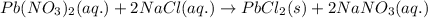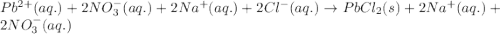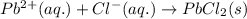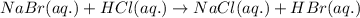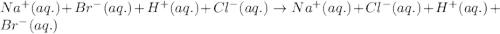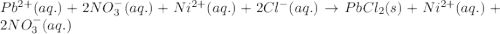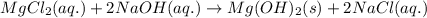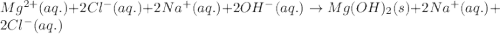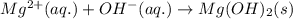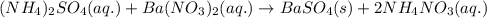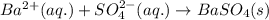Chemistry, 15.10.2019 19:30 davgre1271
Determine net ionic equations, if any, occuring when aqueous solutions of the following reactants are mixed. select "true" or "false" to indicate whether or not the stated reaction (or "no reaction") correctly corresponds to the expected observation in each case. lead(ii) nitrate and sodium chloride; no reaction occurs. sodium bromide and hydrochloric acid; no reaction occurs. nickel(ii) chloride and lead(ii) nitrate; pb2+(aq) + 2cl-(aq) --> pbcl2(s) magnesium chloride and sodium hydroxide; no reaction occurs. ammonium sulfate and barium nitrate; ba2+(aq) + so42-(aq) --> baso4(s)

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:30
How do i complete this electrolysis of water lab? i’m at home, so i don’t have the materials, and the lab didn’t properly work and was incomplete at school.
Answers: 1

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3

Chemistry, 22.06.2019 18:00
How many moles of oxygen gas are produced from the decomposition of six moles of potassium
Answers: 1

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1
You know the right answer?
Determine net ionic equations, if any, occuring when aqueous solutions of the following reactants ar...
Questions

Physics, 16.12.2019 12:31

Social Studies, 16.12.2019 12:31

Chemistry, 16.12.2019 12:31


Physics, 16.12.2019 12:31

Mathematics, 16.12.2019 12:31

Mathematics, 16.12.2019 12:31





Mathematics, 16.12.2019 12:31






History, 16.12.2019 12:31