
Chemistry, 15.10.2019 20:00 tejalawatson91
When a chemical reaction is at equilibrium, which of the following statements is always true?
a. the equilibrium constant keq= 1.
b. the ratio of the concentrations of reactants and products equals 1.
c. the rate of the forward reaction is equal to the rate of the backward reaction.
d. all of the choices are correct.

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 02:00
How many moles of magnesium is 3.01 x10^22 atoms of magnesium?
Answers: 1

Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1

Chemistry, 22.06.2019 21:00
The rate constant for the reaction below is 6.2 x 10−5 mol l−1 s −1. if the initial concentration of a is 0.0500 m, what is its concentration after 115 s?
Answers: 1
You know the right answer?
When a chemical reaction is at equilibrium, which of the following statements is always true?
Questions

Mathematics, 21.09.2021 09:40


Social Studies, 21.09.2021 09:40


Mathematics, 21.09.2021 09:40


History, 21.09.2021 09:40





History, 21.09.2021 09:40





Mathematics, 21.09.2021 09:40

Mathematics, 21.09.2021 09:40


Mathematics, 21.09.2021 09:40

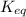
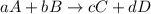
![K_{eq}=\frac{[C]^c[D]^d}{[A]^a[B]^b}](/tpl/images/0322/7004/9c8b0.png)
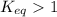 ; the reaction is product favored.When
; the reaction is product favored.When 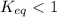 ; the reaction is reactant favored.When
; the reaction is reactant favored.When 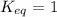 ; the reaction is in equilibrium.
; the reaction is in equilibrium.

