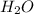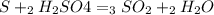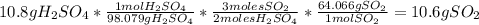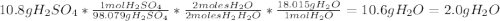Chemistry, 15.10.2019 04:00 johnnyboy41706
For each of the following unbalanced equations, calculate how many grams of each product would be produced by complete reaction of 10.8 g of the second reactant. (a) s(s) + h2so4(aq) → so2(g) + h2o(l) so2 webassign will check your answer for the correct number of significant figures. g h2o webassign will check your answer for the correct number of significant figures. g

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:20
Calcium hydride (cah2) reacts with water to form hydrogen gas: cah2(s) + 2h2o(l) → ca(oh)2(aq) + 2h2(g) how many grams of cah2 are needed to generate 45.0 l of h2 gas at a pressure of 0.995 atm and a temperature of 32 °c?
Answers: 2

Chemistry, 22.06.2019 12:10
|using the periodic tablewarm-upuse the periodic table in the tools bar to answer the following questions.what elemental classification does oxygen belongto? done
Answers: 3

Chemistry, 22.06.2019 16:00
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

You know the right answer?
For each of the following unbalanced equations, calculate how many grams of each product would be pr...
Questions

Mathematics, 25.06.2019 01:00


Biology, 25.06.2019 01:00

History, 25.06.2019 01:00

Mathematics, 25.06.2019 01:00


English, 25.06.2019 01:00

English, 25.06.2019 01:00



Physics, 25.06.2019 01:00

Mathematics, 25.06.2019 01:00



Mathematics, 25.06.2019 01:00

Mathematics, 25.06.2019 01:00

Mathematics, 25.06.2019 01:00



Mathematics, 25.06.2019 01:00

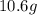 of
of 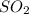
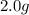 of
of