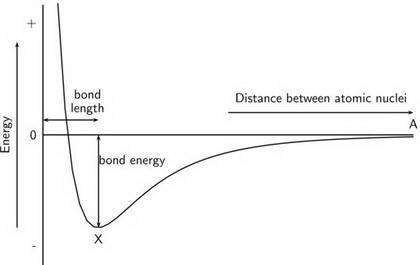
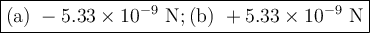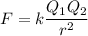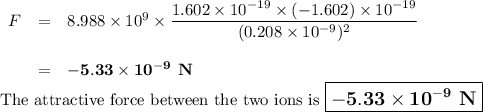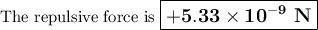Chemistry, 14.10.2019 22:00 tylrmannon
The atomic radii of li+ and 02 ions are 0.068 and 0.140 nm, respectively. (a) calculate the force of attraction between these two ions at their equilibrium interionic separation (i. e., when the ions just touch one another). (b) what is the force of repulsion at this same separation distance? 2.19 for a k+-c- ion pair, attractive and repulsive energies ea and er, respectively, depend on the distance between the ions r, according to

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:30
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 12:00
What are the first two quantum numbers for the electrons located in subshell 4d? what are the first three quantum numbers for the electrons located in subshell 2s? how many electrons can be held in a sublevel l = 3? how many electrons can be held in the energy level n = 4? how many electrons in an atom can share the quantum numbers n = 4 and l = 3?
Answers: 1

Chemistry, 22.06.2019 23:00
If two identical atoms are bonded,what kind of molecule is formed
Answers: 1
You know the right answer?
The atomic radii of li+ and 02 ions are 0.068 and 0.140 nm, respectively. (a) calculate the force of...
Questions







Computers and Technology, 13.03.2020 22:21



Computers and Technology, 13.03.2020 22:21




English, 13.03.2020 22:21



Computers and Technology, 13.03.2020 22:21

Computers and Technology, 13.03.2020 22:21