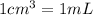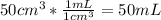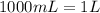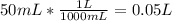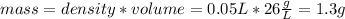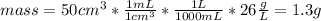Suppose you have 50.0 cm³ of a substance with a density of 26.0 g/l and you want to determine the mass of the substance. the calculation can be broken down into three steps. first, convert the volume from cubic centimeters to milliliters. then, convert the volume from milliliters to liters. finally, determine the mass of the sample in grams. show the unit analysis by placing the correct components into the unit‑factor slots.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:30
What problem would a person have if the nucleic acid in one of his or her cells were damaged?
Answers: 2

Chemistry, 22.06.2019 00:30
Elements that do not have full outer electron shells will donate, share, or take electrons from other atoms. choose the items that have the correct binary ionic formula.
Answers: 2

Chemistry, 22.06.2019 02:00
How many moles of magnesium is 3.01 x10^22 atoms of magnesium?
Answers: 1

Chemistry, 22.06.2019 07:20
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1
You know the right answer?
Suppose you have 50.0 cm³ of a substance with a density of 26.0 g/l and you want to determine the ma...
Questions

Mathematics, 29.08.2019 18:30

Chemistry, 29.08.2019 18:30



Mathematics, 29.08.2019 18:30

History, 29.08.2019 18:30

Mathematics, 29.08.2019 18:30

Mathematics, 29.08.2019 18:30




Mathematics, 29.08.2019 18:30

Chemistry, 29.08.2019 18:30

Mathematics, 29.08.2019 18:30

English, 29.08.2019 18:30



Computers and Technology, 29.08.2019 18:30


History, 29.08.2019 18:30