
Chemistry, 11.10.2019 23:10 erniewernie
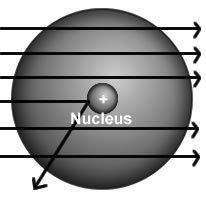
The results of rutherford's gold foil experiment gave him the evidence to arrive at two conclusions: (1) an atom was much more than just empty space and scattered electrons and (2)
a) in any atom, electrons orbit a central nucleus.
b) all atoms were composed of three subatomic particles.
c) an atom must have a positively charged center that contains most of its mass.
d) an atom must have an equal number of positive and negative particles in a central location.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:00
According to the tide table below what time of day will the highest tide occur?
Answers: 1



Chemistry, 22.06.2019 07:00
If there is any 12 to 14 girls that need a boyfriend just follow me and let me know
Answers: 1
You know the right answer?
The results of rutherford's gold foil experiment gave him the evidence to arrive at two conclusions:...
Questions


Advanced Placement (AP), 06.11.2020 23:00


Chemistry, 06.11.2020 23:00




English, 06.11.2020 23:10


History, 06.11.2020 23:10

Mathematics, 06.11.2020 23:10


Mathematics, 06.11.2020 23:10

Arts, 06.11.2020 23:10






English, 06.11.2020 23:10



