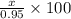Chemistry, 11.10.2019 22:30 kimsouther2
Amixture of kclo3 and kcl with a mass of 0.950 g was heated to produce o2. after heating, the mass of residue was 0.820 g. assuming all the kclo3 decomposed to kcl and o2, calculate the mass percent of kclo3 in the original mixture.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:40
Brainliesttt me asap! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1

Chemistry, 22.06.2019 10:10
When electrolyzing copper (ll) chloride, what reaction takes place at the anode? what reaction takes place at the cathode?
Answers: 1

Chemistry, 22.06.2019 11:20
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1

Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1
You know the right answer?
Amixture of kclo3 and kcl with a mass of 0.950 g was heated to produce o2. after heating, the mass o...
Questions

English, 31.12.2019 11:31


Mathematics, 31.12.2019 11:31

Social Studies, 31.12.2019 11:31



Mathematics, 31.12.2019 11:31

Mathematics, 31.12.2019 11:31

Advanced Placement (AP), 31.12.2019 11:31



Biology, 31.12.2019 11:31




Business, 31.12.2019 11:31

Mathematics, 31.12.2019 11:31



History, 31.12.2019 11:31

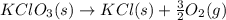
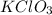 = 122.5 g/mol
= 122.5 g/mol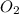 = 32 g/mole
= 32 g/mole
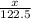
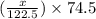
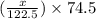 = 0.820 g
= 0.820 g