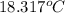Aquantity of 85.0 ml of 0.900 m hcl is mixed with 85.0 ml of 0.900 m koh in a constantpressure calorimeter that has a heat capacity of 325 j/°c. if the initial temperatures of both solutions are the same at 18.24°c, what is the final temperature of the mixed solution? the heat of neutralization is −56.2 kj/mol. assume the density and specific heat of the solutions are the same as those for water.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Match each object to its description: a. coma of a comet b. comet tail c. oort cloud haze surrounding a nucleus created by solar wind. hypothetical sphere around the solar system
Answers: 1

Chemistry, 22.06.2019 03:00
Which step in naming unsaturated hydrocarbons is used for alkenes but not alkynes
Answers: 2

Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2

Chemistry, 22.06.2019 08:30
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3
You know the right answer?
Aquantity of 85.0 ml of 0.900 m hcl is mixed with 85.0 ml of 0.900 m koh in a constantpressure calor...
Questions






English, 01.06.2021 19:00

Mathematics, 01.06.2021 19:00

Computers and Technology, 01.06.2021 19:00

Mathematics, 01.06.2021 19:00



Mathematics, 01.06.2021 19:00

Mathematics, 01.06.2021 19:00

Mathematics, 01.06.2021 19:00


Mathematics, 01.06.2021 19:00


History, 01.06.2021 19:00


Mathematics, 01.06.2021 19:00

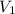 = 85.0 ml,
= 85.0 ml, 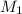 = 0.9 M
= 0.9 M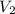 = 85.0 ml,
= 85.0 ml, 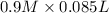 (as 1 L = 1000 ml so, 85 ml = 0.085 L)
(as 1 L = 1000 ml so, 85 ml = 0.085 L)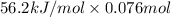
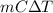
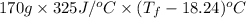
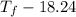
 =
=