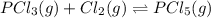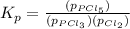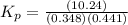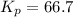Chemistry, 10.10.2019 00:10 GingerSnaps
Phosphorous trichloride and phosphorous pentachloride equilibrate in the presence of molecular chlorine according to the reaction: pcl3 (g) + cl2 (g) → pcl5 (g) an equilibrium mixture at 450 k contains ppcl3 = 0.348 atm, pcl2 = 0.441 atm, and ppcl5 = 10.24 atm. what is the value of kp at this temperature? phosphorous trichloride and phosphorous pentachloride equilibrate in the presence of molecular chlorine according to the reaction: (g) + (g) (g) an equilibrium mixture at 450 k contains = 0.348 atm, = 0.441 atm, and = 10.24 atm. what is the value of kp at this temperature? 66.7 1.50 ⋅ 10−2 12.99 1.57 9.45

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Iwll give extra points to who gets this for ! what type of reaction is this? ?
Answers: 2

Chemistry, 22.06.2019 08:30
Which change in temperature is the smallest? a change of 1 thomson degree a change of 1 kelvin degree a change of 1 fahrenheit degree a change of 1 celsius degree
Answers: 1


You know the right answer?
Phosphorous trichloride and phosphorous pentachloride equilibrate in the presence of molecular chlor...
Questions



Health, 24.01.2020 00:31











Computers and Technology, 24.01.2020 00:31




Chemistry, 24.01.2020 00:31


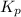 at this temperature is 66.7
at this temperature is 66.7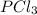 at equilibrium = 0.348 atm
at equilibrium = 0.348 atm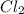 at equilibrium = 0.441 atm
at equilibrium = 0.441 atm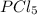 at equilibrium = 10.24 atm
at equilibrium = 10.24 atm