
Chemistry, 09.10.2019 03:30 fancycar14
Pentaborane-9, b5h9, is a colorless, highly reactive liquid that will burst into flame when exposed to oxygen. the reaction is 2b5h9(l) + 12o2(g) ⟶ 5b2o3(s) + 9h2o(l) calculate the kilojoules of heat released per gram of the compound reacted with oxygen. the standard enthalpy of formation of b5h9 is 73.2 kj/mol.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Rocks, as they are compressed, begin forming mountains above the earth's surface when two continental plates converge. the continental crust increases in depth as the mountains grow above. the himalayan mountains formed at a convergent plate boundary in this manner. the rocks are smashed together causing them to due to the intense heat and pressure from the colliding plates and eventually forming rock. a) melt; igneous b) layer; sedimentary c) recrystallize; metamorphic d) melt into the earth's interior; metamorphic
Answers: 1

Chemistry, 22.06.2019 10:30
Earth's axis of rotation is tilted at an angle of 23.5 degrees. what is one change you would see on earth if its axis was not tilted?
Answers: 3


Chemistry, 23.06.2019 01:00
Heat energy, carbon dioxide, and water are released through which process? a. photosynthesis b. depolymerization c. digestion d. cellular respiration
Answers: 1
You know the right answer?
Pentaborane-9, b5h9, is a colorless, highly reactive liquid that will burst into flame when exposed...
Questions

Mathematics, 22.04.2020 17:53



Mathematics, 22.04.2020 17:53


Mathematics, 22.04.2020 17:53




Mathematics, 22.04.2020 17:53

Mathematics, 22.04.2020 17:53

Mathematics, 22.04.2020 17:53



English, 22.04.2020 17:53


Arts, 22.04.2020 17:53


History, 22.04.2020 17:53

Geography, 22.04.2020 17:53

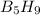 is -71.92 kJ
is -71.92 kJ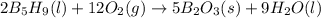
![\Delta H^o_{rxn}=\sum [n\times \Delta H^o_f_{(product)}]-\sum [n\times \Delta H^o_f_{(reactant)}]](/tpl/images/0302/4270/72c39.png)
![\Delta H^o_{rxn}=[(5\times \Delta H^o_f_{(B_2O_3(s))})+(9\times \Delta H^o_f_{(H_2O(l))})]-[(2\times \Delta H^o_f_{(B_5H_9(l))})+(12\times \Delta H^o_f_{(O_2(g))})]](/tpl/images/0302/4270/e310e.png)
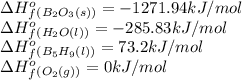
![\Delta H^o_{rxn}=[(5\times (1271.94))+(9\times (-285.83))]-[(2\times (73.2))+(12\times (0))]\\\\\Delta H^o_{rxn}=-9078.57kJ](/tpl/images/0302/4270/015ae.png)
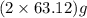 of
of 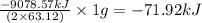 of energy.
of energy.

