
Chemistry, 08.10.2019 21:30 NaVaThEBeAsT
Iron ore can be reduced to iron by the following reaction: fe2o3(s) + 3h2(g) → 2fe + 3h2o(l) (a) how many moles of fe can be made from 1.25 moles of fe2o3? (b) how many moles of h2 are needed to make 3.75 moles of fe? (c) if the reaction yields 12.50 moles of h2o, what mass of fe2o3 was used

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
A100-watt light bulb radiates energy at a rate of 100 j/s. (the watt, a unit of power or energy over time, is defined as 1 j/s.) if all of the light emitted has a wavelength of 525 nm , how many photons are emitted per second?
Answers: 1

Chemistry, 22.06.2019 00:30
Drive down any three characteristic of modern periodic table
Answers: 1

Chemistry, 22.06.2019 07:10
An experimental procedure requires a 10 ml of acid to be dissolved
Answers: 2

You know the right answer?
Iron ore can be reduced to iron by the following reaction: fe2o3(s) + 3h2(g) → 2fe + 3h2o(l) (a) ho...
Questions

Mathematics, 07.04.2021 04:10







Mathematics, 07.04.2021 04:10







Mathematics, 07.04.2021 04:10

Mathematics, 07.04.2021 04:10

Mathematics, 07.04.2021 04:10

Mathematics, 07.04.2021 04:10


Chemistry, 07.04.2021 04:10

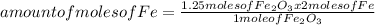
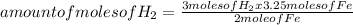
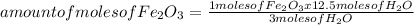
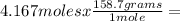 665.4699 grams
665.4699 grams

