Chemistry, 08.10.2019 05:30 karla21pilots
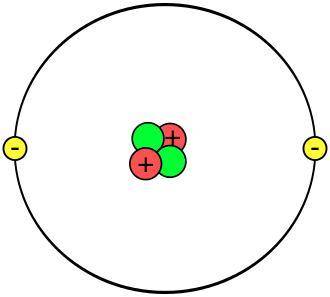
Chapter 2 active reading guide the chemical context of life this chapter covers the basics that you may have learned in your chemistry class. whether your teacher goes over this chapter, or assigns it for you to review on your own, the questions that follow should you focus on the most important points. section 1 1. define and give an example of the following terms: mr. croft term definition matter: element: compound: 2. what four elements make up 96% of all living matter? 3. what is the difference between an essential element and a trace element? section 2 4. sketch a model of an atom of helium, showing the electrons, protons, neutrons, and atomic nucleus.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:10
What approach is required to balance the objectives of sustainable development? balancing the objectives of sustainable development requires a(n) .
Answers: 3

Chemistry, 22.06.2019 06:00
Oxidation-reduction reactions (often called "redox" for short) are reactions that involve the transfer of electrons from one species to another. oxidation states, or oxidation numbers, allow chemists to keep track of these electron transfers. in general, one element will lose electrons (oxidation), with the result that it will increase in oxidation number, and another element will gain electrons (reduction), thereby decreasing in oxidation number. the species that is oxidized is called the reducing agent or reductant. the species that is reduced is called the oxidizing agent or oxidant. to sum up: oxidation = increase in oxidation state = loss of electrons = reducing agent reduction = decrease in oxidation state = gain of electrons = oxidizing agent part a which element is oxidized in this reaction? fe2o3+3co→2fe+3co2 enter the elemental symbol. view available hint(s) is oxidized part b which element is reduced in this reaction? 2hcl+2kmno4+3h2c2o4→6co2+2mno2+2kcl+4h2o enter the elemental symbol. view available hint(s) is reduced
Answers: 1

Chemistry, 22.06.2019 12:30
If 22.5 liters of oxygen reacted with excess of hydrogen, how many liters of water vapor could be produced?
Answers: 3

Chemistry, 22.06.2019 23:30
Match each statement with the state of matter it describes
Answers: 3
You know the right answer?
Chapter 2 active reading guide the chemical context of life this chapter covers the basics that you...
Questions


Chemistry, 06.10.2020 01:01

History, 06.10.2020 01:01



Mathematics, 06.10.2020 01:01

History, 06.10.2020 01:01

Mathematics, 06.10.2020 01:01

English, 06.10.2020 01:01




History, 06.10.2020 01:01

Chemistry, 06.10.2020 01:01