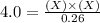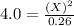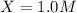Chemistry, 08.10.2019 01:00 hamadehassan
For the equilibrium pcl5(g) pcl3(g) + cl2(g), kc = 4.0 at 228°c. if pure pcl5 is placed in a 1.00-l container and allowed to come to equilibrium, and the equilibrium concentration of pcl5(g) is 0.26 m, what is the equilibrium concentration of pcl3?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 11:00
The number to the right of an element's symbol (ex. c-12) identifies the of an isotope.
Answers: 1

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 22.06.2019 19:50
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

Chemistry, 22.06.2019 20:00
Suppose that some of the compound spilled out of the crucible after it was heated. would that cause the percent by mass of water in the compound determined by the experiment to be too low, too high, or unchanged? briefly explain your answer.
Answers: 1
You know the right answer?
For the equilibrium pcl5(g) pcl3(g) + cl2(g), kc = 4.0 at 228°c. if pure pcl5 is placed in a 1.00-l...
Questions

English, 13.09.2020 22:01

Physics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

World Languages, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Social Studies, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

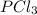 is, 1.0 M
is, 1.0 M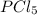 = 0.26 M
= 0.26 M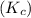 = 4.0
= 4.0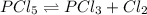
![K_c=\frac{[PCl_3][Cl_2]}{[PCl_5]}](/tpl/images/0298/6790/73fe0.png)
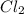 are equal.
are equal.