
Chemistry, 07.10.2019 23:00 vsuescun10
Based upon the ion charge of the following polyatomic ions, predict the formula for the following compounds. sulfate = so4 -2 phosphate = po4 -3 hydroxide = oh- sodium hydroxide na(oh)2 na(oh)3 na2oh naoh

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:00
What is the molar mass of babr2? a. 217.2 g/mol b. 297.1 g/mol c. 354.5 g/mol d. 434.4 g/mol
Answers: 1

Chemistry, 21.06.2019 19:30
Water molecules have a strong attraction to each other because of hydrogen bonding, allowing water to move against gravity up a plant's stem through capillary action. true false
Answers: 2

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3

Chemistry, 22.06.2019 15:20
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3
You know the right answer?
Based upon the ion charge of the following polyatomic ions, predict the formula for the following co...
Questions


History, 23.04.2020 03:10


Social Studies, 23.04.2020 03:10





English, 23.04.2020 03:10

History, 23.04.2020 03:10

Physics, 23.04.2020 03:10



Mathematics, 23.04.2020 03:10


Mathematics, 23.04.2020 03:10

Mathematics, 23.04.2020 03:10



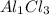
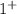 and as OH, the charge is
and as OH, the charge is 
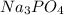 as sodium has
as sodium has 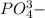 charge.
charge.
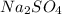 as sodium has
as sodium has 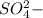 charge.
charge.
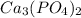 because Calcium possess
because Calcium possess 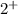 charge and phosphate has
charge and phosphate has 

