
Chemistry, 07.10.2019 01:10 sandlobster6274
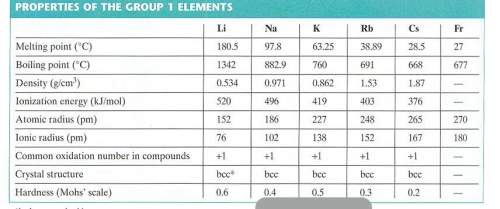
Use the radius of a rubidium atom from the table below to calculate the number of rubidium atoms in a row 6.00 cm long. assume that each rubidium atom touches the ones next to it.
atoms?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:30
Astudent is trying to create a table that compares hypotheses, theories, and laws. hypothesis theory law do scientific researchers formulate it? yes yes yes does it explain why things happen? yes yes no yes yes yes is it used to make predictions? no yes yes which of the following questions would most likely fill the blank in the table? is it an intelligent guess? is it newly formulated? is it based on observations? has it been proved?
Answers: 1

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3

Chemistry, 22.06.2019 20:30
Draw a line graph showing the relationship between temperature in kelvin as a function of kinetic energy.
Answers: 3

You know the right answer?
Use the radius of a rubidium atom from the table below to calculate the number of rubidium atoms in...
Questions

Mathematics, 21.09.2020 20:01


Mathematics, 21.09.2020 20:01





Mathematics, 21.09.2020 20:01

Mathematics, 21.09.2020 20:01




Mathematics, 21.09.2020 20:01


History, 21.09.2020 20:01

Computers and Technology, 21.09.2020 20:01

History, 21.09.2020 20:01


Spanish, 21.09.2020 20:01




