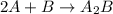Amultistep reaction can only occur as fast as its slowest step. therefore, it is the rate law of the slow step that determines the rate law for the overall reaction. consider the following multistep reaction: a + b → ab (slow) a + ab → a2b (fast)2a + b→ a2b (overall) based on this mechanism, determine the rate law for the overall reaction.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 16:20
When water dissolves sugar, which process is not involved? o dissociation o hydration o surface area of the solute increases sa
Answers: 1

Chemistry, 22.06.2019 16:30
Explain in detail of the four major scientific developments that spurred the formulation of the plate tectonics theory
Answers: 2


Chemistry, 23.06.2019 01:30
If a particle has z = 25 and 23 electrons, what is its charge?
Answers: 2
You know the right answer?
Amultistep reaction can only occur as fast as its slowest step. therefore, it is the rate law of the...
Questions

Mathematics, 02.09.2019 00:50


Mathematics, 02.09.2019 00:50

Mathematics, 02.09.2019 00:50


Mathematics, 02.09.2019 00:50


Geography, 02.09.2019 00:50




History, 02.09.2019 00:50



Mathematics, 02.09.2019 00:50

Biology, 02.09.2019 00:50




![Rate=k[A][B]](/tpl/images/0288/1503/27e48.png)
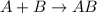 (slow)
(slow)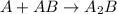 (fast)
(fast)