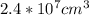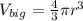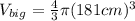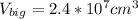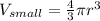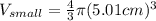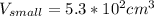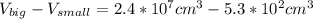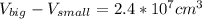Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Given sno2 + 2h2 - sn + 2h20 tin oxide reacts with hydrogen to produce tin and water. how many moles of sno2 are needed to produce 500.0 grams of sn?
Answers: 3

Chemistry, 22.06.2019 09:00
An excess of lithium oxide undergoes a synthesis reaction with water to produce lithium hydroxide li2o+h2o→2lioh if 1.05 g of water reacted, what is the theoretical yield of lithium hydroxide? a) 5.83 x 10–2 g lioh b) 1.17 x 10–1 g lioh c) 2.79 x 100 g lioh d) 1.40 x 100 g lioh
Answers: 1

Chemistry, 22.06.2019 15:40
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

Chemistry, 22.06.2019 22:30
Amedication is given at a dosage of 3.000 mg of medication per kg of body weight. if 0.1500 g of medication is given, then what was the patient's weight in pounds (lbs)? there are 453.59g in 1 lb.
Answers: 2
You know the right answer?
One sphere has a radions of 181 cm, another has a radius of 5.01 cm. what is the difference in volum...
Questions

Mathematics, 31.08.2021 14:10


Mathematics, 31.08.2021 14:10

Chemistry, 31.08.2021 14:10

Mathematics, 31.08.2021 14:10

Geography, 31.08.2021 14:10

World Languages, 31.08.2021 14:10

Social Studies, 31.08.2021 14:10

Mathematics, 31.08.2021 14:10

Chemistry, 31.08.2021 14:10


Chemistry, 31.08.2021 14:10


Social Studies, 31.08.2021 14:10

Mathematics, 31.08.2021 14:10

Mathematics, 31.08.2021 14:10

Health, 31.08.2021 14:10

World Languages, 31.08.2021 14:10

Mathematics, 31.08.2021 14:10