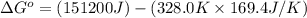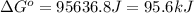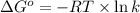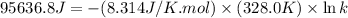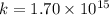For the reaction fe3o4(s) + 4h2(g) --> 3fe(s) + 4h2o(g)
h° = 151.2 kj and s° = 169.4...

Chemistry, 02.10.2019 21:30 bluebabyyy
For the reaction fe3o4(s) + 4h2(g) --> 3fe(s) + 4h2o(g)
h° = 151.2 kj and s° = 169.4 j/k
the equilibrium constant for this reaction at 328.0 k is

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 13:30
A48 g piece of ice at 0.0 ∘c is added to a sample of water at 7.4 ∘c. all of the ice melts and the temperature of the water decreases to 0.0 ∘c. how many grams of water were in the sample?
Answers: 1



Chemistry, 22.06.2019 12:30
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1
You know the right answer?
Questions




Business, 12.07.2019 12:40

Biology, 12.07.2019 12:40




History, 12.07.2019 12:40

Business, 12.07.2019 12:40

Social Studies, 12.07.2019 12:40


History, 12.07.2019 12:40

Social Studies, 12.07.2019 12:40

History, 12.07.2019 12:40


Biology, 12.07.2019 12:40

History, 12.07.2019 12:40



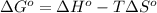
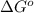 = standard Gibbs free energy = ?
= standard Gibbs free energy = ?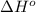 = standard enthalpy = 151.2 kJ = 151200 J
= standard enthalpy = 151.2 kJ = 151200 J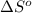 = standard entropy = 169.4 J/K
= standard entropy = 169.4 J/K