
Chemistry, 02.10.2019 22:00 hunterwilliams375
What is the vapor pressure at 23 oc of a solution of 1.20 g of naphthalene, c10h8, in 25.6 g of benzene, c6h6? . the vapor pressure of pure benzene at 23 oc is 86.0 mm hg; the vapor pressure of naphthalene can be neglected. calculate the vapor-pressure lowering of the solution.

Answers: 3
Another question on Chemistry


Chemistry, 21.06.2019 23:30
Two atoms interact with each other as shown by the equation. complete the equation by filling in the missing parts. 1 2 3 4 5 h he li
Answers: 2

Chemistry, 22.06.2019 04:00
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 10:00
Miner's coal distributors does not mine coal itself, nor does it even store or handle the coal. instead, miner's solicits orders for low sulfur coal from other firms, then purchases the required amount from suppliers and directs them to ship the coal to its customers. what is miner's
Answers: 1
You know the right answer?
What is the vapor pressure at 23 oc of a solution of 1.20 g of naphthalene, c10h8, in 25.6 g of benz...
Questions

Mathematics, 11.01.2021 15:20

Mathematics, 11.01.2021 15:20





Biology, 11.01.2021 15:20

English, 11.01.2021 15:20

Mathematics, 11.01.2021 15:20

Mathematics, 11.01.2021 15:20





Mathematics, 11.01.2021 15:20


History, 11.01.2021 15:20



Mathematics, 11.01.2021 15:20

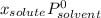 (1)
(1)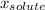 is molar fraction of solute
is molar fraction of solute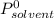 is the capor pressure of the pure solvent (86,0 mmHg)
is the capor pressure of the pure solvent (86,0 mmHg)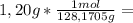 9,36x10⁻³ moles solute
9,36x10⁻³ moles solute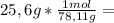 0,328 moles solvent
0,328 moles solvent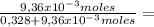 0,0278
0,0278

