
Chemistry, 02.10.2019 21:00 gorbyalexis
Calculate the equilibrium concentrations of n2o4 and no2 at 25 ∘c in a vessel that contains an initial n2o4 concentration of 0.0654 m . the equilibrium constant kc for the reaction n2o4(g)⇌2no2(g) is 4.64×10−3 at 25 ∘c.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:10
Which is true of transition metals when moving from left to right on the periodic table? the d sublevels are not filled across the period. the cation radii become larger across the period. atomic radii increase slightly and then start to decrease. atomic radii decrease slightly and then start to increase. o
Answers: 2

Chemistry, 22.06.2019 05:50
According to coulomb's law, how would the electrical force between particles change if the product of their electrical charge increased?
Answers: 1

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 3

Chemistry, 23.06.2019 01:00
An unsaturated hydrocarbon is a hydrogen-carbon compound with a. a network solid structure b. single bonds c. single bonds in a branched-chain structure d. double or triple bonds
Answers: 1
You know the right answer?
Calculate the equilibrium concentrations of n2o4 and no2 at 25 ∘c in a vessel that contains an initi...
Questions

Mathematics, 21.10.2019 18:20

Mathematics, 21.10.2019 18:20

Computers and Technology, 21.10.2019 18:20

Mathematics, 21.10.2019 18:20




Mathematics, 21.10.2019 18:20

Mathematics, 21.10.2019 18:20

Arts, 21.10.2019 18:20





Mathematics, 21.10.2019 18:20


Mathematics, 21.10.2019 18:20

Mathematics, 21.10.2019 18:20


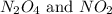 are 0.0164 M and 0.0572 M
are 0.0164 M and 0.0572 M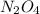 = 0.0654 M
= 0.0654 M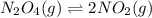
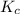 for above reaction follows:
for above reaction follows:![K_c=\frac{[NO_2]_{eq}^2}{[N_2O_4]_{eq}}](/tpl/images/0284/1377/77dc1.png)
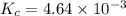
![[NO_2]_{eq}=2x](/tpl/images/0284/1377/0bbcc.png)
![[N_2O_4]_{eq}=0.0654-x](/tpl/images/0284/1377/db879.png)
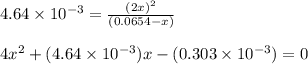
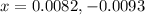
![[NO_2]_{eq}=2x=2(0.0082)=0.0164M](/tpl/images/0284/1377/53c5f.png)
![[N_2O_4]_{eq}=0.0654-x=(0.0654-0.0082)=0.0572M](/tpl/images/0284/1377/125d4.png)


