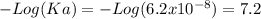Chemistry, 02.10.2019 20:20 keving4three
Aphosphate buffer is involved in the formation of urine. the developing urine contains h2po4 and hpo42- in the same concentration as present in blood plasma. identify the acid and its conjugate base. write the ionization equation and the ka expression. 6.2 x 10^(8) a. the ka of h2po4 is 6.2 x 108. what is the pka? is this acid weaker or stronger than h2co3? which buffer system is more optimal for regulating ph in the body? b. using lechatlier's principle, what happens if the urine is acidified (h+ ions added)?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Solar energy is energy from the sun that is converted into thermal or energy. a. nuclear b. mechanical c. electrical d. chemical
Answers: 2



Chemistry, 22.06.2019 18:00
What amount of heat is exchanged when 106.2 grams of substance y goes from a liquid at 35 degrees celsius to a solid at the same temperature? melting point of substance y = 35 degrees c; δhvaporization = 3.67 j/mol; δhfusion = 3.30 j/mol. mwsubstance y = 28.22 g/mol. −12.4 j −3.51 x 102 j 1.24 x 101 j 351 j
Answers: 1
You know the right answer?
Aphosphate buffer is involved in the formation of urine. the developing urine contains h2po4 and hpo...
Questions


Mathematics, 30.03.2021 15:30

Mathematics, 30.03.2021 15:30


Chemistry, 30.03.2021 15:30

Mathematics, 30.03.2021 15:30



Mathematics, 30.03.2021 15:30

Social Studies, 30.03.2021 15:30

Chemistry, 30.03.2021 15:30



Mathematics, 30.03.2021 15:30


English, 30.03.2021 15:30




Mathematics, 30.03.2021 15:30

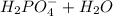 ⇄
⇄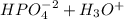 (1)
(1)![Ka = \frac{[HPO_{4}^{-2}] [H_{3}O^{+}]}{[H_{2}PO_{4}^{-}] [H_{2}O]} = 6.2x10^{-8}](/tpl/images/0284/0159/77441.png)