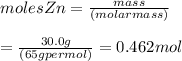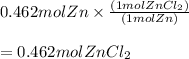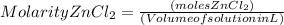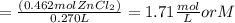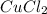Chemistry, 01.10.2019 22:30 yousifgorgees101
What is the molarity of zncl2 that forms when 30.0 g of zinc completely reacts with cucl2 according to the following reaction? assume a final volume of 270 ml . zn(s)+cucl2(aq)→zncl2(aq)+cu(s)

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Y=‐1x + 7 if y has a value of ‐24 what is the value of x?
Answers: 1

Chemistry, 22.06.2019 19:30
Which one of the following substances would be the most soluble in ccl4? na2so4 h2o ch3ch2ch2ch2oh c4h10 hi
Answers: 1

Chemistry, 22.06.2019 20:00
In vapor-liquid equilibrium in a binary mixture, both components are generally present in both phases. how many degrees of freedom are there for such a system? the reaction between nitrogen and hydrogen to form ammonia occurs in the gas phase. how many degrees of freedom are there for this system? steam and coal react at high temperatures to form hydrogen, carbon monoxide, carbon dioxide, and methane. the following reactions have been suggested as being involved in the chemical transformation:
Answers: 3

You know the right answer?
What is the molarity of zncl2 that forms when 30.0 g of zinc completely reacts with cucl2 according...
Questions

Mathematics, 02.10.2020 17:01


English, 02.10.2020 17:01



History, 02.10.2020 17:01


Social Studies, 02.10.2020 17:01



History, 02.10.2020 17:01


English, 02.10.2020 17:01

Mathematics, 02.10.2020 17:01


Mathematics, 02.10.2020 17:01

English, 02.10.2020 17:01



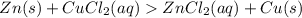
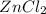 ( using mole ratio 1 : 1 )
( using mole ratio 1 : 1 )