
Chemistry, 01.10.2019 20:30 anikabarnhart
Consider the following reaction representing the combustion of propane: + o2 → co2 + h2o a. balance the equation b. how many moles of oxygen are required to burn 1 mol of propane? c. how many grams of oxygen are required to burn 100 g of propane? d. at standard temperature and pressure, what volume of oxygen would be required to burn 100 g of propane? if air is 21 percent oxygen, what volume of air at stp would be required? e. at stp, what volume of carbon dioxide would be produced when 100 g of propane are burned? f. the standard enthalpy of propane is -103.8 kj/mol. find the gross heat released when 1 kg is burned.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 05:00
Frictional forces acting on an object are often converted into energy, which causes the temperature of the object to rise slightly.
Answers: 2

Chemistry, 22.06.2019 08:30
Which metal exist in liquid state and can be cut with knife ?
Answers: 2

Chemistry, 22.06.2019 13:30
Which is true of a liquid? it has a definite volume but not a definite mass.it has a definite mass but not a definite volume.it has a definite volume but not a definite shape.it has a definite shape but not a definite volume.
Answers: 2

You know the right answer?
Consider the following reaction representing the combustion of propane: + o2 → co2 + h2o a. balanc...
Questions

Mathematics, 16.12.2019 14:31



Physics, 16.12.2019 14:31

Chemistry, 16.12.2019 14:31

Physics, 16.12.2019 14:31

History, 16.12.2019 14:31

History, 16.12.2019 14:31

Advanced Placement (AP), 16.12.2019 14:31

Physics, 16.12.2019 14:31

Mathematics, 16.12.2019 14:31


History, 16.12.2019 14:31

Physics, 16.12.2019 14:31


Chemistry, 16.12.2019 14:31


Biology, 16.12.2019 14:31


History, 16.12.2019 14:31

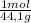 ×
×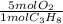 ×
×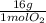 = 181 g of O₂
= 181 g of O₂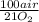 = 1210 L of air
= 1210 L of air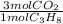 = 6,80 moles of CO₂
= 6,80 moles of CO₂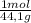 = 22,68 moles
= 22,68 moles

