Background info:
the standard enthalpy of formation (δh∘f) is the enthalpy change that occurs...

Chemistry, 19.08.2019 10:30 terryonsavage543
Background info:
the standard enthalpy of formation (δh∘f) is the enthalpy change that occurs when exactly1 mol of a compound is formed from its constituent elements under standard conditions. the standard conditions are 1 atm pressure, a temperature of 25 ∘c , and all the species present at a concentration of 1 m . a "standard enthalpies of formation table" containing δh∘f values might look something like this: substanceδh∘fh(g)218 kj/molh2(g)0 kj/molba(s)0 kj/molba2+(aq)−538.4 kj/molc(g)71 kj/molc(s)0 kj/moln(g)473 kj/molo2(g)0 kj/molo(g)249 kj/mols2(g)129 kj/mol
question:
what is the balanced chemical equation for the reaction used to calculate δh∘f of baco3(s)? if fractional coefficients are required, enter them as a fraction (i. e. 1/3). indicate the physical states using the abbreviation (s), (l), or (g) for solid, liquid, or gas, respectively. use (aq) for aqueous solution.
express answer as a chemical equation. explain for me !

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 13:10
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1

Chemistry, 22.06.2019 14:00
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1

Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 3
You know the right answer?
Questions

Arts, 14.12.2019 03:31







Mathematics, 14.12.2019 03:31


Mathematics, 14.12.2019 03:31

Mathematics, 14.12.2019 03:31

History, 14.12.2019 03:31

Mathematics, 14.12.2019 03:31

Geography, 14.12.2019 03:31


Social Studies, 14.12.2019 03:31

Mathematics, 14.12.2019 03:31

Mathematics, 14.12.2019 03:31

English, 14.12.2019 03:31

Computers and Technology, 14.12.2019 03:31

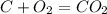
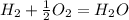


 , using hydrogen as a catalyst.
, using hydrogen as a catalyst.

