
Chemistry, 28.09.2019 03:10 Tonyang1742
Calculate as° for the reaction below: n2(g)+202(g) 2no2(g) where aso for n2(g), o2(g), & no2(g), respectively, is 191.5, 205.0, & 240.5 j/mol-k -156.0 j/k 156.0 j/k 120.5 j/k -120.5 j/k o ooo

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 12:00
1. if you have a gas at 127 degrees c, what is it's absolute temperature (kelvin)? a. 200kb. 300kc. 400kd. 500k2. if you had a gas whose absolute temperature measured 45 k, what is that temperature in celsius? a. -228 cb. -300 cc. 125 cd. 112 c
Answers: 2

Chemistry, 22.06.2019 13:00
12. calculate the hydroxide ion concentration of a solution with ph = 3.25. show all calculations leading to an answer
Answers: 3

Chemistry, 22.06.2019 14:20
Which statement explains why the bonds between non metals tend to be covalent? the bonds are found to be nondirectional they have large differences in electronegativity they have small differences in electronegativity they have ions that produce an electrostatic pull
Answers: 1
You know the right answer?
Calculate as° for the reaction below: n2(g)+202(g) 2no2(g) where aso for n2(g), o2(g), & no2(g...
Questions

Biology, 16.09.2019 12:30


History, 16.09.2019 12:30

Mathematics, 16.09.2019 12:30

Mathematics, 16.09.2019 12:30

Mathematics, 16.09.2019 12:30




Mathematics, 16.09.2019 12:30

Business, 16.09.2019 12:30

Mathematics, 16.09.2019 12:30





Chemistry, 16.09.2019 12:30



English, 16.09.2019 12:30

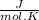 - [ 1 mol × 191,5
- [ 1 mol × 191,5 

