
Chemistry, 27.09.2019 07:20 amselah3571
Cs2(g) + o2(g) co2(g) + so2(g)
consider the unbalanced equation above. a 0.150 g sample of cs2 is reacted with 285 ml of o2 at stp. what is the total volume of the final mixture of gases at stp?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 13:00
Identify the balanced chemical equation that represents a decomposition reaction. p4 + 3o2 ⟶ p2o3 2fe(oh)3 ⟶ 2feo3 + h2o cuso4 ⟶ cuo + 2so3 2fe(oh)3 ⟶ fe2o3 + 3h2o
Answers: 1

Chemistry, 22.06.2019 09:00
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

Chemistry, 22.06.2019 13:30
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3

Chemistry, 22.06.2019 18:00
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2
You know the right answer?
Cs2(g) + o2(g) co2(g) + so2(g)
consider the unbalanced equation above. a 0.150 g sample...
consider the unbalanced equation above. a 0.150 g sample...
Questions


Mathematics, 14.12.2020 07:50

History, 14.12.2020 07:50



English, 14.12.2020 07:50




English, 14.12.2020 07:50


Mathematics, 14.12.2020 08:00

World Languages, 14.12.2020 08:00

Mathematics, 14.12.2020 08:00

Mathematics, 14.12.2020 08:00

Mathematics, 14.12.2020 08:00



Mathematics, 14.12.2020 08:00

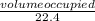
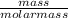
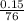 = 0.00197mole
= 0.00197mole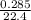 = 0.01272mole
= 0.01272mole

