
Chemistry, 27.09.2019 03:30 Knownothing
Ethyl iodide (c2h5i) decomposes at a certain temperature in the gas phase as follows: c2h5i(g) → c2h4(g) + hi(g) from the following data determine the order of the reaction and the rate constant. time (min) [c2h5i] (m) 0.0 2.00 15.0 1.82 30.0 1.64 48.0 1.42 75.0 1.10

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 19:00
Iknow the answer to 13 is b and 14 is d. i just need to know why the correct answers are correct
Answers: 3

Chemistry, 21.06.2019 23:00
Why are the trends and exceptions to the trends in ionization energy observed?
Answers: 1

Chemistry, 22.06.2019 02:20
Compared with the freezing-point depression of a 0.01 m c6h12o6 solution, the freezing-point depression of a 0.01 m nacl solution is
Answers: 1

Chemistry, 22.06.2019 10:00
Ill give brainiestif one neutron initiates a fission event that produces two neutrons in the products, how many new reactions can now be initiated? if each of the neutrons produced in the first fission event then initiates a fission event that produces one neutron in the products, how many new reactions can now be initiated by each neutron? how many neutrons in total were produced by the two fission events described?
Answers: 2
You know the right answer?
Ethyl iodide (c2h5i) decomposes at a certain temperature in the gas phase as follows: c2h5i(g) → c2...
Questions


English, 05.03.2021 22:10

Chemistry, 05.03.2021 22:10







Engineering, 05.03.2021 22:10

Mathematics, 05.03.2021 22:10

Mathematics, 05.03.2021 22:10

English, 05.03.2021 22:10


Mathematics, 05.03.2021 22:10





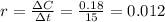
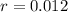 .
.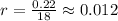
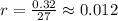
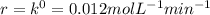 that is, the reaction is the zero order respect to C2H5I since it is not depending on concentration of C2H5I.
that is, the reaction is the zero order respect to C2H5I since it is not depending on concentration of C2H5I. 


