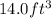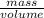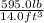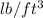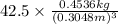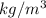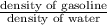Chemistry, 25.09.2019 02:30 leslcookie23
Asample of gasoline has a mass of 595.0 lb and a volume of 14.0 ft^3. what is its specific gravity?

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

Chemistry, 22.06.2019 04:00
Acontainer holds 35.8 moles of gas under 10.0 atm of pressure at 70.0 c. what is the volume of the container?
Answers: 2

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2
You know the right answer?
Asample of gasoline has a mass of 595.0 lb and a volume of 14.0 ft^3. what is its specific gravity?...
Questions













Social Studies, 11.02.2020 21:39




Biology, 11.02.2020 21:40