
Chemistry, 25.09.2019 02:20 lexizamora2
1.00 kg of ice at -10 °c is heated using a bunsen burner flame until all the ice melts and the temperature reaches 95 °c. a) how much energy in kj is required to effect this transformation?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:30
Asolution of sodium hydroxide was titrated against a solution of sulfuric acid. how many moles of sodium hydroxide would react with 1 mole of sulfuric acid?
Answers: 2

Chemistry, 22.06.2019 07:20
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1

Chemistry, 22.06.2019 08:30
In the reaction between a crushed antacid tablet and vinegar what gas is emitted
Answers: 2

Chemistry, 22.06.2019 14:00
What term describes technology that operates on an atomic level
Answers: 2
You know the right answer?
1.00 kg of ice at -10 °c is heated using a bunsen burner flame until all the ice melts and the tempe...
Questions


Mathematics, 10.12.2021 16:10

Mathematics, 10.12.2021 16:10


Mathematics, 10.12.2021 16:10

Social Studies, 10.12.2021 16:10


History, 10.12.2021 16:10


Mathematics, 10.12.2021 16:10


Mathematics, 10.12.2021 16:10


Biology, 10.12.2021 16:10

SAT, 10.12.2021 16:10

Engineering, 10.12.2021 16:10




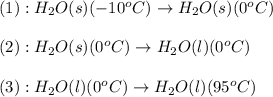
![\Delta H=[m\times c_{p,s}\times (T_{final}-T_{initial})]+n\times \Delta H_{fusion}+[m\times c_{p,l}\times (T_{final}-T_{initial})]](/tpl/images/0260/0438/5cd06.png)
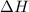 = energy required = ?
= energy required = ?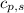 = specific heat of solid water =
= specific heat of solid water = 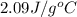
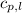 = specific heat of liquid water =
= specific heat of liquid water = 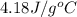
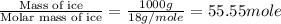
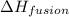 = enthalpy change for fusion = 6.01 KJ/mole = 6010 J/mole
= enthalpy change for fusion = 6.01 KJ/mole = 6010 J/mole![\Delta H=[1000g\times 4.18J/gK\times (0-(-10))^oC]+55.55mole\times 6010J/mole+[1000g\times 2.09J/gK\times (95-0)^oC]](/tpl/images/0260/0438/a7596.png)
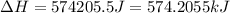 (1 KJ = 1000 J)
(1 KJ = 1000 J)


