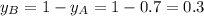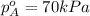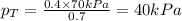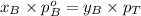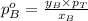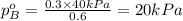Components a and b form ideal solution. at 350 k, a liquid mixture containing 40% (mole) a is in equilibrium with a vapour containing 70% (mole) a. if the vapour pressure of a at 350 k is 70 kpa, what is the vapour pressure of b? (b) 20 kpa (d) 12 kpa (а) 25 kpa (c) 40 kpa

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:40
Ted and emily played a mixed doubles tennis match against jack and brenda. in the second match. ted and brenda played against jack and emily. which type of chemical reaction does the situation demonstrate?
Answers: 3

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3


Chemistry, 23.06.2019 03:00
The size (radius) of an oxygen molecule is about 2.0 ×10−10m. make a rough estimate of the pressure at which the finite volume of the molecules should cause noticeable deviations from ideal-gas behavior at ordinary temperatures (t= 300k ). assume that deviatons would be noticeable when volume of the gas per molecule equals the volume of the molecule itself.
Answers: 3
You know the right answer?
Components a and b form ideal solution. at 350 k, a liquid mixture containing 40% (mole) a is in equ...
Questions



Chemistry, 21.11.2019 05:31

Biology, 21.11.2019 05:31

Mathematics, 21.11.2019 05:31




English, 21.11.2019 05:31


Biology, 21.11.2019 05:31


Social Studies, 21.11.2019 05:31



Mathematics, 21.11.2019 05:31

Social Studies, 21.11.2019 05:31

English, 21.11.2019 05:31


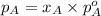 .............(1)
.............(1)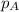 = partial vapor pressure of A
= partial vapor pressure of A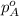 = vapor pressure of pure substance A
= vapor pressure of pure substance A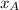 = mole fraction of A
= mole fraction of A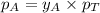 .............(2)
.............(2)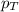 = total pressure of the mixture
= total pressure of the mixture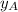 = mole fraction of A
= mole fraction of A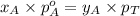
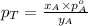 ............(3)
............(3)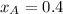 and
and 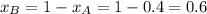
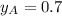 and
and