Chemistry, 24.09.2019 23:20 maskythegamer
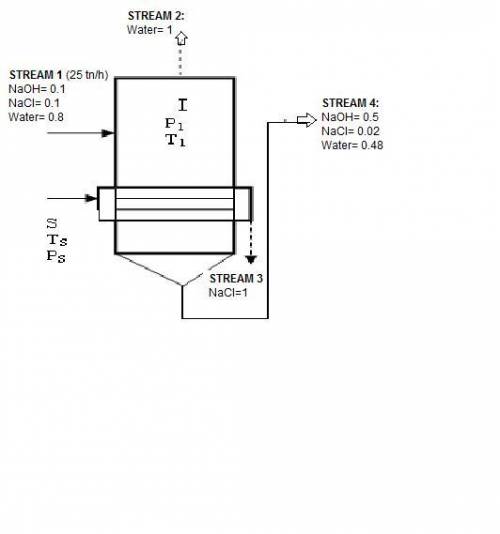
In an evaporator 25 ton / h of a solution of 10% naoh, 10% nacl, and 80% water by weight. during evaporation, the water evaporates and the salt precipitates like crystals they are allowed to settle and are removed. the outgoing concentrated solution of the evaporator contains 50% naoh, 2% nacl and 48% water. based on this information is requested:
1. draw the process flow diagram, indicating each of its streams and compositions (known and unknown).
2. calculate the kilograms of precipitated salt and the kilograms of solution concentrated for every hour of work.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 19:30
The molecular formula for caffeine is cshion402. which of the following elements is not found in caffeine?
Answers: 1

Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1

Chemistry, 22.06.2019 15:00
Which are forms of frozen water? check all that apply. dew frost hail rain sleet
Answers: 2

Chemistry, 22.06.2019 22:30
Write and balance the chemical equation that represents the reaction of aqueous sulfuric acid with aqueous sodium hydroxide to form water and sodium sulfate. include phases.
Answers: 1
You know the right answer?
In an evaporator 25 ton / h of a solution of 10% naoh, 10% nacl, and 80% water by weight. during eva...
Questions

Mathematics, 10.12.2021 23:20



English, 10.12.2021 23:20


Mathematics, 10.12.2021 23:20


Mathematics, 10.12.2021 23:20


History, 10.12.2021 23:20

Mathematics, 10.12.2021 23:20



Mathematics, 10.12.2021 23:20

Arts, 10.12.2021 23:20




Health, 10.12.2021 23:20