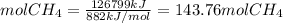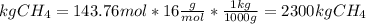The reaction is as follows: ch4 + 202 + co2 + 2h2o the enthalpy of reaction between methane ( 2 ) and oxygen ( 2 ) is exothermic (-882 kj/mol) with respect to the dissipation of methane. it is of interest to generate 126799 kj of energy from the combustion. how many kg of methane will need to be combusted? molecular weight: c-12 kg/kmol h-1 kg/kmol 0 - 16 kg/kmol select one: o a. 2.300 o b. 2300.209 o c. 1.725 o d. 0.009

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:00
How many moles of magnesium is 3.01 x10^22 atoms of magnesium?
Answers: 1

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 23.06.2019 00:00
Predict the relative bond lengths of the three carbon-oxygen bonds in the carbonate ion (co2−3). what would you expect the charge to be on each oxygen? match the words in the left column to the appropriate blanks in the sentences on the right. make certain each sentence is complete before submitting your answer.
Answers: 3

Chemistry, 23.06.2019 00:00
Which is true about metals used for jewelry, such as platinum and gold? a. they have low flammability. b. they have low reactivity. c. they have high flammability. d. they have high reactivity.
Answers: 1
You know the right answer?
The reaction is as follows: ch4 + 202 + co2 + 2h2o the enthalpy of reaction between methane ( 2 ) a...
Questions





Mathematics, 24.07.2019 03:40

Mathematics, 24.07.2019 03:40

Computers and Technology, 24.07.2019 03:40

Mathematics, 24.07.2019 03:40

History, 24.07.2019 03:40


Chemistry, 24.07.2019 03:40


Physics, 24.07.2019 03:40

Chemistry, 24.07.2019 03:40

History, 24.07.2019 03:40

Mathematics, 24.07.2019 03:40

Health, 24.07.2019 03:50

Mathematics, 24.07.2019 03:50

Mathematics, 24.07.2019 03:50