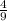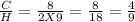Acombustion reaction involves the reaction of a substance with oxygen gas. the complete combustion of any hydrocarbon (binary compound of carbon and hydrogen) produces carbon dioxide and water as the only products. octane is a hydrocarbon that is found in gasoline. complete combustion of octane produces 8 l of carbon dioxide for every 9 l of water vapor (both measured at the same temperature and pressure). what is the ratio of carbon atoms to hydrogen atoms in a molecule of octane?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 1

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3


Chemistry, 22.06.2019 22:00
If a solution contains 3 moles/liter of sodium chloride (nacl, made of sodium ions and chloride ions), what is the osmolarity of this solution
Answers: 3
You know the right answer?
Acombustion reaction involves the reaction of a substance with oxygen gas. the complete combustion o...
Questions




Mathematics, 02.09.2021 21:10

Health, 02.09.2021 21:10

Mathematics, 02.09.2021 21:10

Computers and Technology, 02.09.2021 21:10


History, 02.09.2021 21:10

History, 02.09.2021 21:10


Mathematics, 02.09.2021 21:10



Mathematics, 02.09.2021 21:10



English, 02.09.2021 21:10