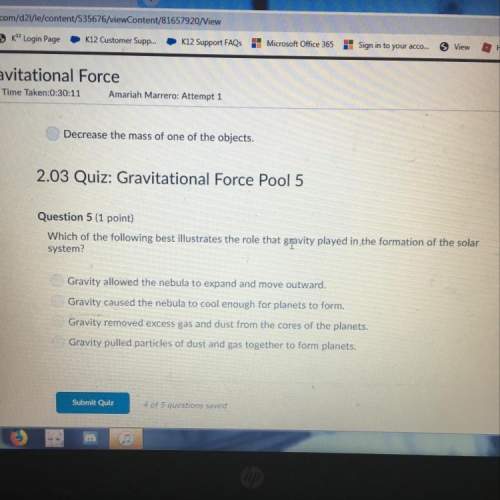
An unidentified organic compound x, containing c, h, and o was subjected to combustion analysis. when 228.4 mg of pure x was burned 627.4 mg of co2 and 171.2 mg of h2o were obtained. determine the simplest formula of the compound x. 1. c5h8o2 2. c6h10o2 3. c10h8o2 4. c6h8o 5. c2h4o

Answers: 2
Another question on Chemistry


Chemistry, 22.06.2019 05:30
According to periodic trend, which of the following most likely has the highest ionization energy? kr be ni sc
Answers: 3

Chemistry, 22.06.2019 05:30
Which other elements contain the same number of outer electrons as sodium
Answers: 3

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1
You know the right answer?
An unidentified organic compound x, containing c, h, and o was subjected to combustion analysis. whe...
Questions

Spanish, 19.01.2021 22:20


Mathematics, 19.01.2021 22:20





Mathematics, 19.01.2021 22:20

Physics, 19.01.2021 22:20

Mathematics, 19.01.2021 22:20

Advanced Placement (AP), 19.01.2021 22:20


Mathematics, 19.01.2021 22:20


Mathematics, 19.01.2021 22:20




English, 19.01.2021 22:20

Mathematics, 19.01.2021 22:20