
Valproic acid, used to treat seizures and bipolar disorder, is composed of c, h, and o. a 0.165-g sample is combusted to produce 0.166 g of water and 0.403 g of carbon dioxide. what is the empirical formula for valproic acid? if the molar mass is 144 g> mol, what is the molecular formula?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 1


Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1

Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2
You know the right answer?
Valproic acid, used to treat seizures and bipolar disorder, is composed of c, h, and o. a 0.165-g sa...
Questions





History, 18.03.2021 18:50

Biology, 18.03.2021 18:50






Health, 18.03.2021 18:50

Mathematics, 18.03.2021 18:50


Mathematics, 18.03.2021 18:50


Chemistry, 18.03.2021 18:50

History, 18.03.2021 18:50

Social Studies, 18.03.2021 18:50

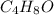
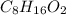
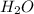 = 0.166 g /18 g/mol = 0.00922 moles
= 0.166 g /18 g/mol = 0.00922 moles
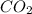 = 0.403 g /44.01 g/mol = 0.009157 moles
= 0.403 g /44.01 g/mol = 0.009157 moles


