
Chemistry, 19.09.2019 04:30 friendsalwaysbae
Homework 3 write the balanced equations. calculate how many grams of each reactant will be needed to obtain 100.0 grams of the insoluble product formed in the reaction. show complete solutions. 1. aluminum chloride + calcium hydroxide aluminum hydroxide + calcium chloride 2. mercury (ii) oxide mercury + oxygen 3. barium nitrate + copper (ii) sulfate barium sulfate+ copper (ii) nitrate 4. lead (ii) chloride + potassium iodide lead (ii) iodide + potassium chloride 5. sodium sulfide + copper (ii) chloride copper (ii) sulfide + sodium chloride

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
An electrons position cannot be known precisely only it's probability of being in a certain location can be known
Answers: 1


Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 14:00
In the space, show a correct numerical setup for calculating the number of moles of co2 present in 11 grams of co2
Answers: 1
You know the right answer?
Homework 3 write the balanced equations. calculate how many grams of each reactant will be needed to...
Questions


Mathematics, 03.03.2021 18:20

Mathematics, 03.03.2021 18:20

Mathematics, 03.03.2021 18:20



History, 03.03.2021 18:20



Mathematics, 03.03.2021 18:20



Social Studies, 03.03.2021 18:20



Computers and Technology, 03.03.2021 18:20

Mathematics, 03.03.2021 18:20



Mathematics, 03.03.2021 18:20

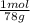 = 1,282 moles of Al(OH)₃.
= 1,282 moles of Al(OH)₃.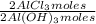 = 1,282 moles of AlCl₃ ×
= 1,282 moles of AlCl₃ × 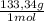 = 170,9 g of AlCl₃
= 170,9 g of AlCl₃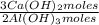 = 1,923 moles of Ca(OH)₂ ×
= 1,923 moles of Ca(OH)₂ × 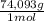 = 142,5 g of Ca(OH)₂
= 142,5 g of Ca(OH)₂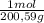 = 0,4985 moles of Hg.
= 0,4985 moles of Hg.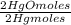 = 0,4985 moles of HgO ×
= 0,4985 moles of HgO × 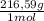 = 108,0 g of HgO
= 108,0 g of HgO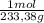 = 0,4285 moles of BaSO₄.
= 0,4285 moles of BaSO₄.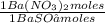 = 0,4285 moles of Ba(NO₃)₂ ×
= 0,4285 moles of Ba(NO₃)₂ × 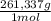 = 112,0 g of Ba(NO₃)₂
= 112,0 g of Ba(NO₃)₂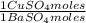 = 0,4285 moles of CuSO₄ ×
= 0,4285 moles of CuSO₄ × 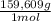 = 68,39 g of CuSO₄
= 68,39 g of CuSO₄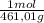 = 0,2169 moles of PbI₂.
= 0,2169 moles of PbI₂.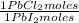 = 0,2169 moles of PbCl₂ ×
= 0,2169 moles of PbCl₂ × 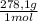 = 60,32 g of PbCl₂
= 60,32 g of PbCl₂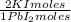 = 0,4338 moles of KI ×
= 0,4338 moles of KI × 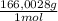 = 72,01 g of KI
= 72,01 g of KI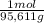 = 1,046 moles of CuS.
= 1,046 moles of CuS.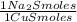 = 1,046 moles of Na₂S ×
= 1,046 moles of Na₂S × 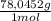 = 81,63 g of Na₂S
= 81,63 g of Na₂S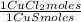 = 1,046 moles of CuCl₂ ×
= 1,046 moles of CuCl₂ × 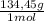 = 140,6 g of CuCl₂
= 140,6 g of CuCl₂

