
Asolution contains 0.1 (10-7) moles of hydroxyl ions (oh-) per liter. which of the following best describes this solution? a solution contains 0.1 (10-7) moles of hydroxyl ions (oh-) per liter. which of the following best describes this solution? basic: h+ donor basic: h+ acceptor acidic: h+ acceptor neutral acidic: h+ donor

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 20:40
Astudent made the lewis dot diagram of a compound shown. what is the error in the lewis dot diagram? a)an o atom should transfer all of its six electrons to mg because the formula is mgo b) both electrons of mg should be transferred to one o adam because the formula is mgo c) the electrons should be transferred from each o add him to capital mg because mg has fewer electrons d) the number of dots around mg should be four because it has to transfer two electrons to each o
Answers: 1

Chemistry, 21.06.2019 23:10
Nitrogen (n), phosphorus (p), and potassium (k) are the main nutrients in plant fertilizers. according to an industry convention, the numbers on the label refer to the mass percents of n, p2o5, and k2o, in that order. calculate the n: p: k ratio of a 30: 10: 10 fertilizer in terms of moles of each element, and express it as x: y: 1.0.
Answers: 1

Chemistry, 22.06.2019 06:00
What type of electromagnetic radiation has a shorter wavelength than blue light
Answers: 2

Chemistry, 22.06.2019 07:00
How many moles are in 7.2 x 10^23 carbon molecules? (*round to the nearest hundredth and include the unit "mol c" after your number) question 6 options:
Answers: 2
You know the right answer?
Asolution contains 0.1 (10-7) moles of hydroxyl ions (oh-) per liter. which of the following best de...
Questions


Computers and Technology, 01.07.2019 07:30



History, 01.07.2019 07:30

Computers and Technology, 01.07.2019 07:30

Health, 01.07.2019 07:30


Social Studies, 01.07.2019 07:30


Mathematics, 01.07.2019 07:30

Physics, 01.07.2019 07:30

English, 01.07.2019 07:30

Social Studies, 01.07.2019 07:30

Mathematics, 01.07.2019 07:30

English, 01.07.2019 07:30

English, 01.07.2019 07:30

Mathematics, 01.07.2019 07:30

Social Studies, 01.07.2019 07:30

Chemistry, 01.07.2019 07:30

![pH=-\log [H^+]](/tpl/images/0241/8360/37e81.png)
![pOH=-\log[OH^-]](/tpl/images/0241/8360/fe336.png)
![pOH=-\log[10^{-7}]](/tpl/images/0241/8360/492ed.png)
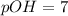
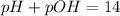
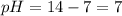
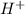 nor donates
nor donates 

