
Chemistry, 18.09.2019 04:10 maddie7155
At elevated temperatures, dinitrogen pentoxide decomposes to nitrogen dioxide and oxygen: 2n2o5(g) → 4no2 (g) + o2 (g) when the rate of formation of no2 is 5.5 ⋅ 10-4 m/s, the rate of decomposition of n2o5 is m/s. at elevated temperatures, dinitrogen pentoxide decomposes to nitrogen dioxide and oxygen: 2n2o5(g) 4no2 (g) + o2 (g) when the rate of formation of no2 is 5.5 10-4 m/s, the rate of decomposition of n2o5 is m/s. 2.8 ⋅ 10-4 1.4 ⋅ 10-4 5.5 ⋅ 10-4 10.1 ⋅ 10-4 2.2 ⋅ 10-3

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:00
Hey guys so i need to know what is _nh3+> nh4oh ~chemistry~
Answers: 1

Chemistry, 22.06.2019 14:00
Will mark brainliest how many electrons can be held in the energy level n = 4?
Answers: 1


Chemistry, 23.06.2019 17:00
During which of the following phases of the moon do we see the left half of the moon as lit? full moon first quarter moon gibbous moon third quarter moon any is greatly : )
Answers: 1
You know the right answer?
At elevated temperatures, dinitrogen pentoxide decomposes to nitrogen dioxide and oxygen: 2n2o5(g)...
Questions



English, 20.06.2020 20:57


Mathematics, 20.06.2020 20:57

English, 20.06.2020 20:57




Mathematics, 20.06.2020 20:57

Mathematics, 20.06.2020 20:57

Biology, 20.06.2020 20:57

Computers and Technology, 20.06.2020 20:57


Biology, 20.06.2020 20:57

Chemistry, 20.06.2020 20:57

Mathematics, 20.06.2020 20:57

Mathematics, 20.06.2020 20:57


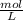 -
-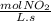 ×
× 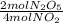 = 2,8 M/s
= 2,8 M/s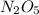 is
is 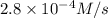
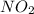 =
= 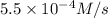
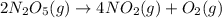
![\text{Rate of decomposition of }N_2O_5=-\frac{1}{2}\times \frac{d[N_2O_5]}{dt}](/tpl/images/0238/6091/c0940.png)
![\text{Rate of formation of }NO_2=+\frac{1}{4}\frac{d[NO_2]}{dt}](/tpl/images/0238/6091/2ef8c.png)
![\text{Rate of formation of }O_2=+\frac{d[O_2]}{dt}](/tpl/images/0238/6091/db5e4.png)
![\text{Rate of reaction}=-\frac{1}{2}\times \frac{d[N_2O_5]}{dt}=+\frac{1}{4}\frac{d[NO_2]}{dt}=+\frac{d[O_2]}{dt}](/tpl/images/0238/6091/52065.png)
![-\frac{1}{2}\times \frac{d[N_2O_5]}{dt}=+\frac{1}{4}\frac{d[NO_2]}{dt}](/tpl/images/0238/6091/5fd92.png)
![-\frac{d[N_2O_5]}{dt}=+\frac{2}{4}\frac{d[NO_2]}{dt}](/tpl/images/0238/6091/ff940.png)
![-\frac{d[N_2O_5]}{dt}=\frac{2}{4}\times (5.5\times 10^{-4}M/s)](/tpl/images/0238/6091/9cde4.png)
![-\frac{d[N_2O_5]}{dt}=2.8\times 10^{-4}M/s](/tpl/images/0238/6091/65aeb.png)


