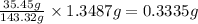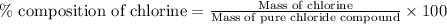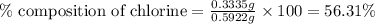Chemistry, 18.09.2019 03:30 shortcake8047
A0.5922 g sample of a pure soluble chloride compound is dissolved in water, and all of the chloride ion is precipitated as agcl by the addition of an excess of silver nitrate. the mass of the resulting agcl is found to be 1.3487 g. what is the mass percentage of chlorine in the original compound?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 05:30
According to periodic trend, which of the following most likely has the highest ionization energy? kr be ni sc
Answers: 3

Chemistry, 22.06.2019 09:00
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 22.06.2019 19:40
What is the wavelength of a 3*10^12 hz infrared wave a 3*10^20m b 1* 10^4m c 3*10^-3m d 1*10^-4 m
Answers: 1
You know the right answer?
A0.5922 g sample of a pure soluble chloride compound is dissolved in water, and all of the chloride...
Questions

Chemistry, 08.01.2021 20:30


Mathematics, 08.01.2021 20:30

Mathematics, 08.01.2021 20:30

Mathematics, 08.01.2021 20:30



Biology, 08.01.2021 20:30







Mathematics, 08.01.2021 20:30

Social Studies, 08.01.2021 20:30



Mathematics, 08.01.2021 20:30

Mathematics, 08.01.2021 20:30