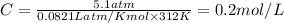The osmotic pressure exerted by a solution is equal to the molarity multiplied by the absolute temperature and the gas constant r. suppose the osmotic pressure of a certain solution is measured to be 5.1 atm at an absolute temperature of 312 k. write an equation that will let you calculate the molarity of this solution. your equation should contain only symbols. be sure you define each symbol other than r .

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 16:30
At 20°c, a sample of h2o liquid and a sample of co2 gas each have the same average kinetic energy. why is one a liquid and the other a gas at this temperature?
Answers: 1

Chemistry, 23.06.2019 04:10
Two solids are mixed in a flask and stirred. after a few minutes, the flask becomes cold. which of the following best describes this reaction? a. an exothermic reaction b. a combustion reaction c. an endothermic reaction d. a decomposition reaction
Answers: 1

Chemistry, 23.06.2019 08:00
Pl what kind of reaction is this? nahco3 + h2o → co2 + naoh + h2o -composition -decomposition -single replacement -double replacement im leaning more toward single replacement. if im wrong can you explain whyy?
Answers: 1

Chemistry, 23.06.2019 09:30
Which of the following is not a characteristic of a hydrogen bond? 1. it is responsible for the unusual physical properties of water. 2. it is weaker than a covalent bond. 3. it is stronger than other dipole-dipole interactions. 4. it can occur when hydrogen is covalently bound to very electronegative elements liks f, cl, br and i.
Answers: 1
You know the right answer?
The osmotic pressure exerted by a solution is equal to the molarity multiplied by the absolute tempe...
Questions







History, 17.10.2020 19:01

Social Studies, 17.10.2020 19:01

Computers and Technology, 17.10.2020 19:01




Chemistry, 17.10.2020 19:01

Mathematics, 17.10.2020 19:01


Mathematics, 17.10.2020 19:01

Mathematics, 17.10.2020 19:01



Physics, 17.10.2020 19:01

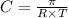

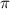 = osmotic pressure = 5.1 atm
= osmotic pressure = 5.1 atm